BIO 102 MENU
syllabus 
1 - origin 
2 - biomol. 
3 - biomol2 
4 - viruses
5 - prokaryon 
6 - endosym 
7 - eukaryon 
8 - energy 
9 - mitosis 
10 - meiosis 
11 - reprod 
12 - genetics 
13 - humgene 
14 - humge2 
15 - evolution 
16 - evolutio2 
17 - diversity 
18 - diversi2 
19 - tissues 
20 -digestive 
21 - respirat 
22 - circul 
23 - excret 
24 - endocr 
25 - receptors 
26 - nervsys 
Quizzes 
Bio 103 Lab 
(full title of lecture appears in status bar on the top or at the bottom of your window)
Biology 102 - General Biology
Energy Metabolism, Acquiring and Releasing Energy (ATP)
Photosynthesis, Glycolysis, Krebs Cycle, Electron Transport Phosphorylation
Metabolism is the sum total of chemical reactions occurring in cells. It includes both anabolism, the synthesis of the biomolecules (e.g., protein synthesis, DNA replication, glucose synthesis in plants) and catabolism, the degradation of molecules usually for the production of energy (e.g., glycolysis, Krebs Cycle). Metabolism is carried out by specific enzymes which catalyze each step of a long series of reactions. The steps in a metabolic pathway may be linear such as those in glycolysis or cyclic as in the Krebs Cycle or light independent reactions of photosynthesis. Each of the thousands of enzymes required for the thousands of metabolic pathways are coded for in our genes. Each enzyme is specific for its substrates and each produces unique products. Some metabolic pathways occur in all or most cells and some are specific to the cell type.
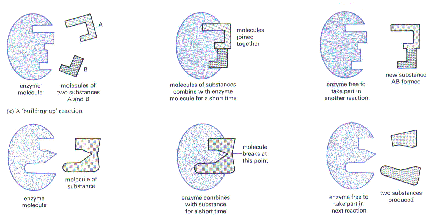
Enzymes can put molecules together (synthesize) or break molecules apart (degrade)
The Role of Enzymes
The enzymes that carry out all metabolic reactions are protein catalysts. They speed up the rates of reactions by lowering the energy of activation of the substrate molecules which they convert to product molecules. Enzymes are proteins and, therefore, are composed of specific sequences of amino acids which, in turn, are coded for by genes. Proteins usually have a globular shape and they have "pockets" into which their substrates fit. In order to carry out their specific reaction, they change their shape to put "stress" on the bonds they are to break or the pockets can bring parts of the substrate molecules into the position to react with one another. Because enzymes are proteins, they are affected by temperature and pH (acidity). Both temperature and pH affect the structure of the protein and, therefore, their function. High temperatures and extreme acid base changes denature (inactivate) them, they lose their specific shape and pockets and can no longer do their job. Most enzymes work best at neutral pH such as that found in the cytosol. However, some proteins work best in acid environments. Pepsin, a digestive enzyme, functions in the acid environment of your stomach and the lysosomal enzymes work only within the acid environment of the lysosome. The lysosomal enzymes are inactivated if the lysosomal membrane is broken and the enzymes are released into the cytosol. An example of the effect of temperature is the pigment pattern of the Siamese cat and Himalayan rabbit. These animals have darker fur on their extremities because the enzyme that makes the dark pigment works only at the cooler temperatures found at the animals' extremities. The enzyme is denatured (inactivated) at the higher temperatures found in the other parts of the animals' bodies.
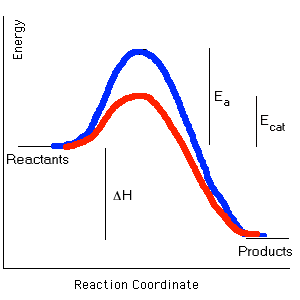
This is an enzyme called a dehydrogenase...it removes H atoms from substrates
All the energy that is required for the synthesis and maintenance of living cells ultimately comes from the sun. The producers (autotrophs) are those organisms which can synthesize their own biomolecules and enough for the consumers, too. The consumers (heterotrophs) such as us and other animals, rely on the metabolic capabilities of the producers. Thus, the photosynthetic organisms, mainly plants, supply all of the basic biomolecules. The recyclers, the fungi and bacteria, break down (degrade) the biomolecules of dead organisms to carbon dioxide, water, and ammonia,which are then used by the producers to make more biomolecules.
Photosynthesis
All organisms require "food" for two reasons: to use as subunits to build the biomolecules of the organism and to provide energy (ATP) for the maintenance and activities of the organism. Energy in the form of ATP is a requirement for all cellular activities. ATP is produced in the light reactions of photosynthesis (in chloroplasts of photosynthetic eukaryotic organisms), in glycolysis (in the cytosol of all organisms), and in aerobic respiration which includes the Krebs Cycle (matrix of mitochondria and cytoplasm of prokaryotic cells) and electron transport phosphorylation (in the mitochondria of eukaryotic organisms and on the inner plasma membrane of prokaryotic cells).
Photosynthesis can be summarized in the equation:
6H2O (water) + 6CO2(carbon dioxide)------->(using light energy)
-------> 6O2 (oxygen) + C6H12O6 (glucose)
The energy of the sun is captured by pigments found in photosynthetic organisms and the energy is captured in such a way as to produce ATP and reducing power in the form of electrons which are carried by the coenzyme, NADPH (formed from the vitamin niacin). In the process, the photons split water into free oxygen and the electrons are captured by the coenzyme, NADPH. NADPH is then used in the light independent (dark) reactions of photosynthesis to reduce (add H atoms to) carbon dioxide to form glucose and later other middle sized biomolecules (also known as carbon fixation).
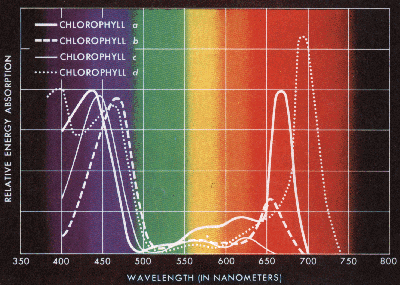
Absorption of visible light by photosynthetic pigments, the chlorophylls

The porphyrin ring in chlorophylls with Mg in the center
(the same ring is in hemoglobin but with Fe in the center)
(see Lecture 1)
The light reactions of photosynthesis occur in the grana (thylakoid membranes) of the chloroplasts of eukaryotic cells. Chlorophyll a captures the blue-violet and red wavelengths of light and chlorophyll b, the blue and red-orange and the accessory pigments capture other wavelengths and pass them to chlorophyll a in the reaction centers. The pigments are parts of a photosystem. In the light reactions, light photons from the sun, literally kick electrons of the pigments up to a higher energy level. These higher energy electrons are passed to chlorophyll a whose electrons are then passed along an electron transport system (in the grana membranes) and the energy of the electrons is captured in the form of ATP, and the electrons and H+ are captured by NADPH. The electrons captured by NADP are replaced by the splitting of water into free oxygen, hydrogen ions and electrons (photolysis). All of the oxygen on this planet comes from the light reactions of photosynthesis.
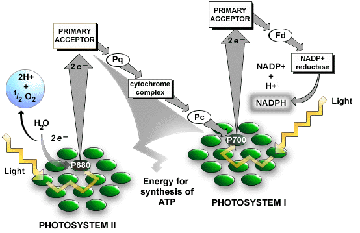
The light-independent reactions of photosynthesis (or dark reactions) can occur in the absence of light. They utilize the products of the light reactions, ATP and NADPH to reduce carbon dioxide and to make glucose and the more complex middle sized biomolecules. The enzymes that carry out these reactions are in the stroma (fluid) portion of the chloroplast. Gas exchange in complex plants occurs through the stomata (open pores) on the underside of the leaves. Carbon dioxide enters and oxygen leaves via these pores.
Glycolysis and the Krebs cycle (aka citric acid cycle) are at the center of metabolism
Aerobic Respiration:
C6H12O6 (glucose) + O2 ---------->
CO2(carbon dioxide) + H2O (water) + ATP (energy)
Glycolysis (1st part of aerobic respiration)
C6H12O6(glucose) --->pyruvate(or lactate or ethanol and CO2)+ATP
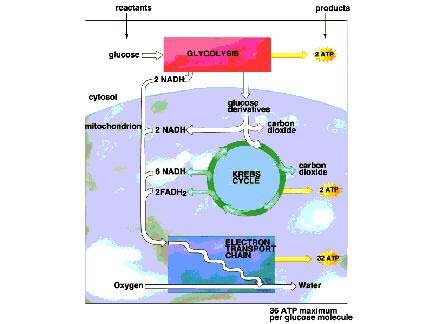
While only some organisms carry out photosynthesis, all cells carry out glycolysis. Glycolysis literally means the breakdown (lysis) of glucose from glycogen. The series of reactions involved in glycolysis is carried out by enzymes that are in the cytosol of all cells, both prokaryotic and eukaryotic cells. Glucose, a six-carbon molecule, is broken down to pyruvate, a three-carbon molecule. A net of 2 ATP molecules is produced per glucose molecule that enters glycolysis. If the cell operates anaerobically, the pyruvate is converted to lactate or ethanol but if the cell is aerobic and oxygen is present, the pyruvate is converted to acetyl CoA. The pyruvate (a 3 carbon molecule) produced from glycolysis is reduced by NADP and after a CO2 is released, the two carbon molecule, acetyl CoA is formed and enters the Krebs Cycle. A total of 36 ATP molecules is produced when glucose undergoes complete oxidation via the Krebs Cycle and is ultimately broken down completely to CO2 and H2O.
Aerobic Respiration: Krebs Cycle and Oxidative Phosphorylation
pyruvate + O2-------------> CO2+ H2O + ATP
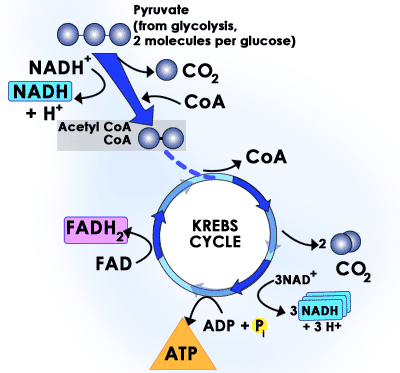
In eukaryotic cells the enzymes of the Krebs Cycle take pyruvate and break it down to CO2 and water. The enzymes that carry out the Krebs Cycle are in the matrix (fluid) portion of the mitochondria. In prokaryotic cells, the Krebs Cycle enzymes are in the cytosol. Two more ATPs are formed as pyruvate is oxidized to carbon dioxide. (The Krebs Cycle, as the name implies is a cyclic series of reactions unlike those of glycolysis which are linear.) In the oxidation of pyruvate through the Krebs Cycle, NADH (a close coenzyme relative of NADPH) is also produced as well as another related coenzyme, FADH. Both these coenzymes carry hydrogen ions (H+) and electrons over to the inner membrane of the mitochondria where electron transport phosphorylation occurs. As the electrons are handed down a series of proteins (cytochromes which contain iron, Fe++), ATP is produced and ultimately, at the end of the line, the electrons are accepted by O2. So the oxygen you breathe is the ultimate electron acceptor in these series of oxidations. The oxygen becomes O- and combines with the H+ to form H2O (metabolic water). In prokaryotic cells, the proteins that perform electron transport phosphorylation are embedded in the inner side of the plasma membrane. This is consistent with the idea that mitochondria originated from aerobic bacteria that were endocytosed.
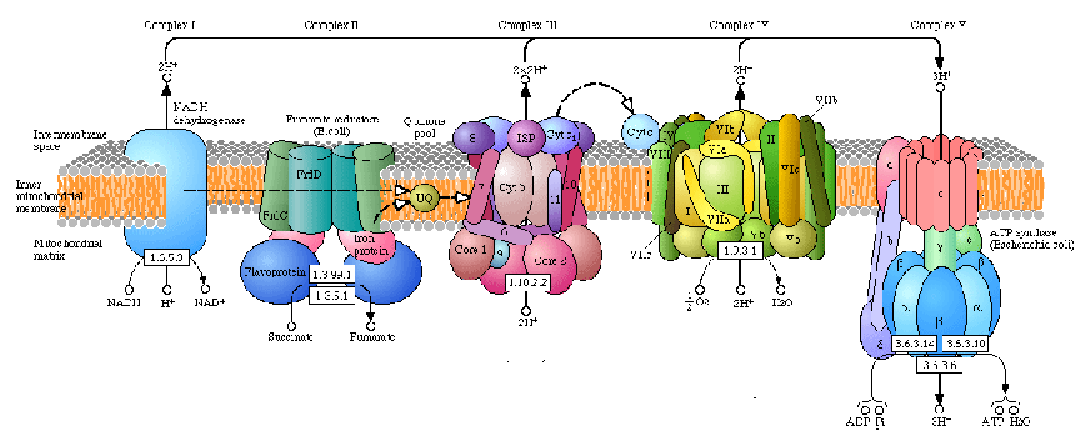
Mitochondrial Electron Transport System

